As our February newsletter has highlighted, collecting real-world oncology data to produce regulatory-compliant and reproducible evidence for your medical research is possible.
The Pulse Platform
Our modular-by-design platform enables you to address your study’s research questions in the following ways:
- By accessing existing de-identified data sets from our ongoing registries, in a secure cloud environment for analysis to occur, utilizing a transparent governance structure.
- By expanding on the existing datasets and adding new variables or instruments such as patient-reported outcomes (PROs) for new data collection efforts.
- By designing a new study or patient registry to collect data to address your specific research question.
Leveraging these Pulse Infoframe capacities will advance your conduct of cutting-edge research and accelerate diagnostic and therapeutic development.
Registry Insights
Real-world data from regulatory-compliant oncology patient Pulse Infoframe registries are now available to researchers and sponsors. Insights from the Global Melanoma Research Network (GMRN), Ocular Melanoma Natural History (OMNi), and Merkel Cell Carcinoma registries are shared below and include novel details about patient diagnosis and treatment effectiveness.
Note: All registry studies are conducted with appropriate informed consent and ethics approval.
GMRN Registry
Real-world data from the GMRN registry were used to investigate immune checkpoint inhibitors in the elderly and determine whether age affected outcomes. Following the analysis of retrospective data from 144 patients, the study concluded that survival rates for patients over the age of 70 were “to be comparable to that achieved in younger cohorts.” To read the full publication, click here.
Additionally, data from the GMRN registry was used to track the real-world experiences of advanced squamous cell carcinoma patients treated with the PD-1 blocker by cemiplamab. To read the full publication, click here.
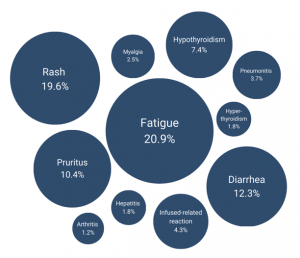
Percentages of adverse reactions reported in the GMRN registry.
OMNi Registry
Real-world data from the OMNi registry were used to better understand how uveal melanoma is managed worldwide by providing crizotinib to high-risk uveal melanoma patients. Using data from the registry, researchers did not notice an overall reduction in cancer relapse. Of the 32 patients enrolled, 9 required dose modified or discontinued because of adverse reactions. To read the full publication, click here.
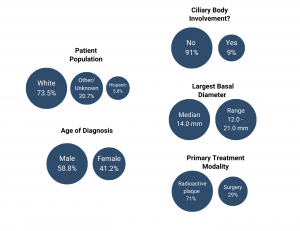
The following graphics showcase insights collected from the study regarding patient demographics and treatment (N=34).
Merkel Cell Carcinoma Registry
Although still in its infancy, the Merkel Cell Carcinoma registry has several publications in progress thanks to the risk real-world data collection facilitated by the committed site investigators and team and the Pulse platform.
How to Learn More and Access Oncology Registries
Please email us at hello@pulseinfoframe.com.